Organic Chemistry Section
Department of Inorganic and Organic Chemistry
Biography
Fèlix Urpí i Tubella is a Full Professor at the Department of Inorganic and Organic Chemistry of the University of Barcelona (UB). Born and raised in Sant Sadurní d'Anoia (Barcelona), he studied at the UB, completing graduate and postgraduate studies in Chemistry. In 1988, under the supervision of Prof. Jaume Vilarrasa, he completed his doctorate. From 1988 to 1989 he carried out postdoctoral research in Prof. David A. Evans' group at Harvard University. After returning to Barcelona, he became a tenured lecturer in the UB's Department of Organic Chemistry in 1991. Since July 2017 he is Full Professor at the Department of Inorganic and Organic Chemistry of the University of Barcelona (UB).
He is member of the UB's Institute of Biomedicine (IBUB).
He was secretary of the Department of Organic Chemistry between November 1996 and January 2000.
He was the Vice-Dean of Research and Doctorate of the Faculty of Chemistry from 2008 until 2017.
Research
Our research activity revolves around the development of wide-ranging, efficient stereoselective processes based on the reactivity of metal enolates and their application to the synthesis of natural products.
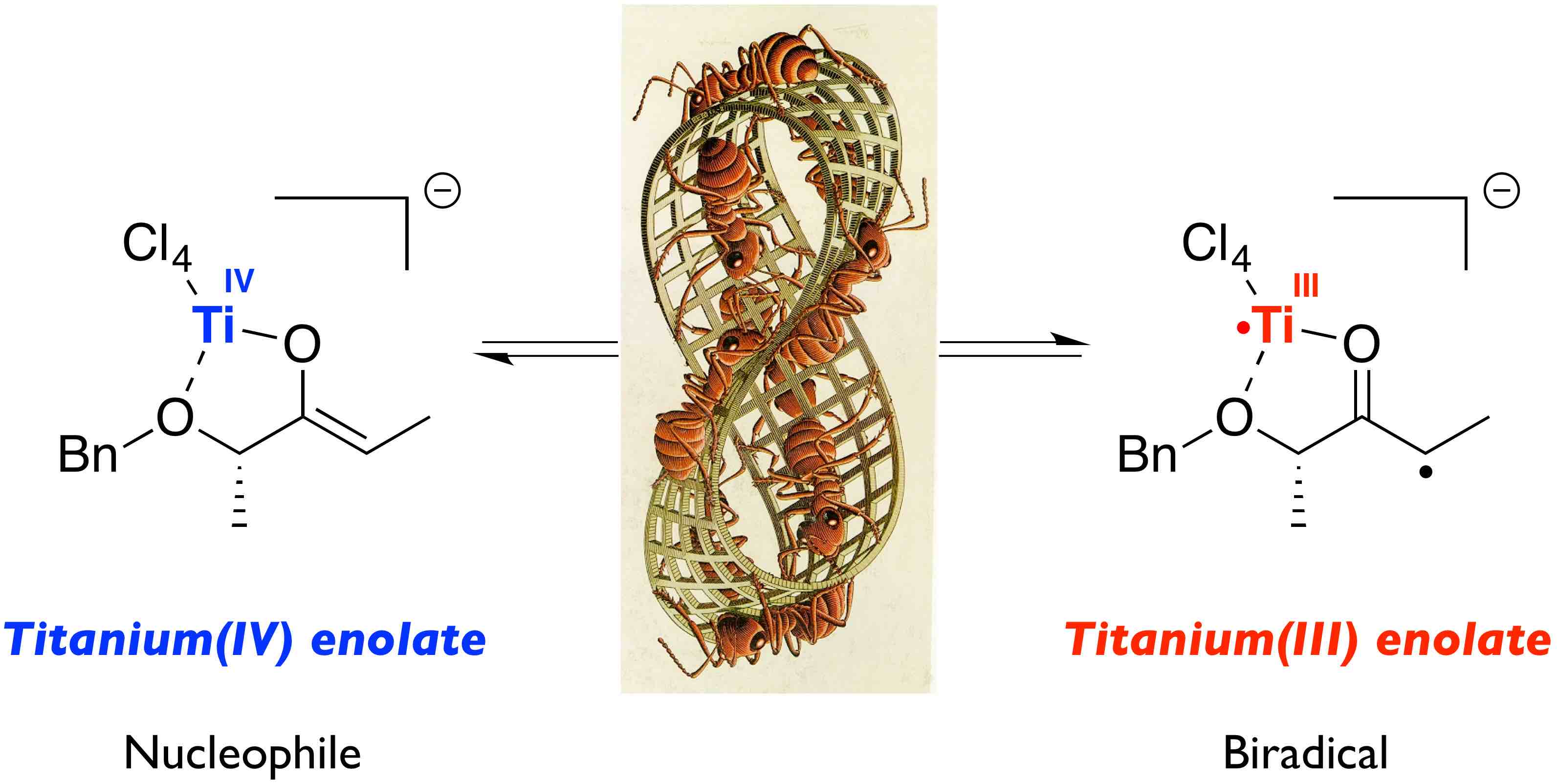
In particular, the high nucleophilicity of titanium(IV) enolates from chiral ketones has been fundamental to the design of new, highly stereoselective Michael and aldol reactions. Additionally, the discovery of their biradical nature has enabled us to use these enolates in radical processes. Essentially, the nucleophilic and the radical nature of titanium(IV) enolates are two aspects of the same species which can be used, without distinction, for the stereoselective construction of carbon-carbon and carbon-oxygen bonds.
We are also interested in the use of structurally simple nickel(II) complexes for the development of new catalytic and asymmetric additions of nickel(II) enolates to cationic intermediates in SN1 processes.
The application of these methods to the synthesis of natural products, such as tedanolide C (pictured on the right), is clear proof of their potential where synthesis is concerned.
Publications
Teaching